- Last Friday, Sorrento Therapeutics appeared to celebrate discovering a cure for the coronavirus in a widely disseminated Fox News article. The company’s stock rose 243% that day on about 78x its daily average volume.
- However, former company employees and experts at Mt. Sinai, the NIH, and the Rockefeller Foundation confirmed to us that the news is likely too good to be true.
- “I would never in my life have put out a press release where I say we have a cure,” a former Sorrento senior executive told us. He also estimated there are “hundreds” of groups with antibodies similar to Sorrento’s.
- “I suggest extreme skepticism regarding any claims made by Sorrento,” another former employee told us.
- “I am not aware of any data suggesting Sorrento’s single Ab is as good as the many others that had already been previously generated (such as ours or Lilly’s)” Regeneron’s co-founder told us.
- Sorrento was nearly out of cash and facing growing questions of solvency leading up to its surprise cure announcement. The company put paperwork in place to sell $500 million of stock just weeks in advance of ‘discovering’ the cure.
- After the financing arrangements were put in place, the company announced a collaboration with Mount Sinai, then within just three days began pitching journalists on its miraculous findings.
- Meanwhile, one Mount Sinai researcher we talked to called Sorrento’s announcement “very hyped”. Another warned us that “nothing in medicine is 100%,” referring to the company’s claim of a solution that works 100% in the Fox News piece.
- One of the company’s financing agreements is with a year-old entity in the British Virgin Islands. The mystery backer was unnamed, but multiple industry sources tell us it is the same individual that was behind financing shipping company DryShips in 2017. DryShips lost 99.9% of its value in 2017 and reverse split eight times between 2016 and 2017.
- We believe that Sorrento’s actions are manipulative at the worst possible time and simply amount to an attempt to shamelessly profiteer off the pandemic.
- We see significant downside from these levels and believe the company is already in the process of severely diluting its new unsuspecting investor base. We believe regulators should closely scrutinize the company’s actions over the last several weeks.
Initial Disclosure: After extensive research, we have taken a short position in shares of Sorrento Therapeutics. We will make any source names and details available to members of the media who wish to independently corroborate our work on condition that they respect their background status. This report represents our opinion, and we encourage every reader to do their own due diligence. Please see our full disclaimer at the bottom of the report.
Introduction: Sorrento Appears to Announce a Complete Cure for COVID-19, Inspiring Hope…and a 243% Gain in Its Stock Price
Last Friday, Sorrento Therapeutics announced, via a Fox News “exclusive”, that it had made a breakthrough in the fight against Covid-19 by discovering an antibody that could imminently “shield the human body from the coronavirus”.
Per the article:
“The company’s CEO said: ‘We want to emphasize there is a cure. There is a solution that works 100 percent,’ Dr. Henry Ji, founder and CEO of Sorrento Therapeutics, told Fox News.”
Alas, after 300,000 COVID-19- related deaths worldwide, millions of confirmed cases and lockdowns across the globe, it finally seemed there was a complete cure. The Fox News piece continued, quoting the CEO:
“If we have the neutralizing antibody in your body, you don’t need the social distancing. You can open up a society without fear.”
It is exactly what everyone in the world wanted to hear.
Predictably, Sorrento’s stock spiked on the news. Prior to the announcement, it had closed at a price of $2.62 and had traded an average of ~6.5 million shares of volume over the preceding 30 days. On the day of the Fox News report, the stock spiked as high as $9 per share, or a gain of 243%, on an astonishing 507 million shares of volume – about 78x its daily average.
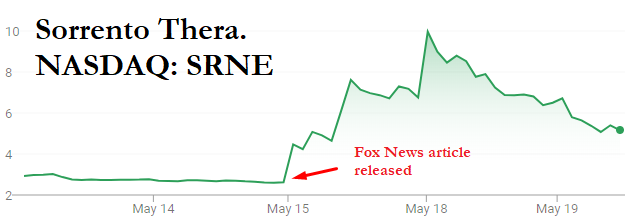
(Source: FactSet pricing data)
But was the news worthy of the company tacking on more than $1 billion in market cap over just several days?
After our research, we certainly don’t think so.
Reality Check: Former Employees, Healthcare Experts from the NIH and Mt. Sinai (A Partner of Sorrento) Speak Out
After speaking with former Sorrento company employees, including those who held senior C-suite and management positions, as well as healthcare experts from outlets like the National Institute of Health and Mt. Sinai (a partner of Sorrento), we came to the conclusion that this “too good to be true” sounding story is exactly that.
We don’t doubt that Sorrento has found the antibody that it claims. One former employee we spoke with said he would be surprised if they couldn’t find the antibody, given their extensive antibody library. Nonetheless, the company remains in the preliminary stages of what we understand to be an arduous process to take its antibody from experiments in vitro (in a petri dish) to a safe and effective treatment in vivo (in humans).
Experts have warned us to take Sorrento’s claims with “extreme skepticism”, saying the announcement seems “very hyped” and with an “aggressive” timeline. One former Sorrento C-suite executive also questioned management’s judgement in putting out a press release stating there was a “cure”.
Additionally, during the course of our research, we uncovered a number of red flags about Sorrento’s past that make us wary not only about the company’s recent claims, but its management team in general.
For example, we found that mere weeks ahead of the ‘cure’ announcement, the company put in place a $250 million financing arrangement with an opaque entity in the British Virgin Islands. The company didn’t disclose the ultimate backer of the entity, but we learned through multiple industry sources that it’s the same individual that helped finance DryShips, a public market disaster which fell 99.9% and did 8 reverse splits between 2016 and 2017.
It seems as though the entire world is fixated on the pandemic right now. This naturally has brought out the best in the human race: people helping others, selfless frontline workers, a scientific community acting in unison to try and solve the global problem.
Unfortunately, it can also bring out the worst. The SEC has already suspended trading in over 25 companies for suspected false or misleading COVID-related claims. This includes two we have recently written about, Predictive Technology Group and SCWorx, which respectively was delisted onto the Grey Sheets (also known hilariously now known as the “Expert Market”) and is in the midst of a regulatory halt.
Basics on Sorrento: A Reverse Merger That Has Spent $375 Million In R&D And Has Amassed A $725 Million Accumulated Deficit
Sorrento came public via reverse merger in 2009 from a shell entity. [Pg. 21]
The company has spent over $375 million on R&D since inception, racking up an accumulated deficit of $725.0 million, but with very little to show for it in terms of commercialized product. [Source, SEC filings & Pg. 3]
The company describes itself as a “clinical stage, antibody-centric” company. Former employees confirmed to us that the company has an extensive antibody library.
The company’s only commercially meaningful product to date is a lidocaine patch called ZTildo. [Pg. 36] Sales of the product have been modest (~$20 million annual revenue run rate) likely owing to substantial competition. Lidocaine has been commercially available since the 1940’s and the product has nearly a dozen direct competitors, including generics.
Activist Short JCapital wrote an exposé on Sorrento in March 2018, which detailed a mass exodus of executives, red flags around its current Chairman/CEO Henry Ji, and the lack of focus of the company. The report also highlighted several examples of company dealings with China-based suspected related parties that have allegedly dissipated the company’s key resources and cash assets.
Given the historical inability to move its extensive R&D efforts into commercialization, and his litany of historical red flags, Sorrento seemed like an unlikely savior in the fight against COVID-19.
The Company Was Nearly Out of Cash and Facing Solvency Questions Before Its “Cure” Announcement
Sorrento was nearly out of cash and facing growing questions of solvency leading up to its surprise cure announcement.
In the company’s latest annual report released in March 2020, it provided investors with a clear warning:
“As a result of our recurring losses from operations, recurring negative cash flows from operations and substantial cumulative losses, there is uncertainty regarding our ability to maintain liquidity sufficient to operate our business effectively, which raises substantial doubt about our ability to continue as a going concern. If we are unsuccessful in our efforts to raise outside financing, we may be required to significantly reduce or cease operations.”
As of the quarter ending March 31st, Sorrento had cash & equivalents of only $21.9 million. The company’s current ratio, a key measure of near-term solvency, was at ~0.55, dangerously below sustainable levels. [Pg. 3] The company’s average quarterly operating cash flow burn rate was $41.7 million in the preceding 4 quarters, indicating that the company was quickly diminishing its remaining resources.
Sorrento Put Paperwork in Place to Sell up to $500 Million In Stock Just Weeks in Advance of ‘Discovering’ The Cure
That same month (March), the company filed a “shelf registration” with the SEC, seeking the ability to offer up to $1 billion in securities. [Pg. 10] It was deemed effective on March 20th.
The company already had a history of issuing significant sums of shares to fund its business, diluting existing shareholders.
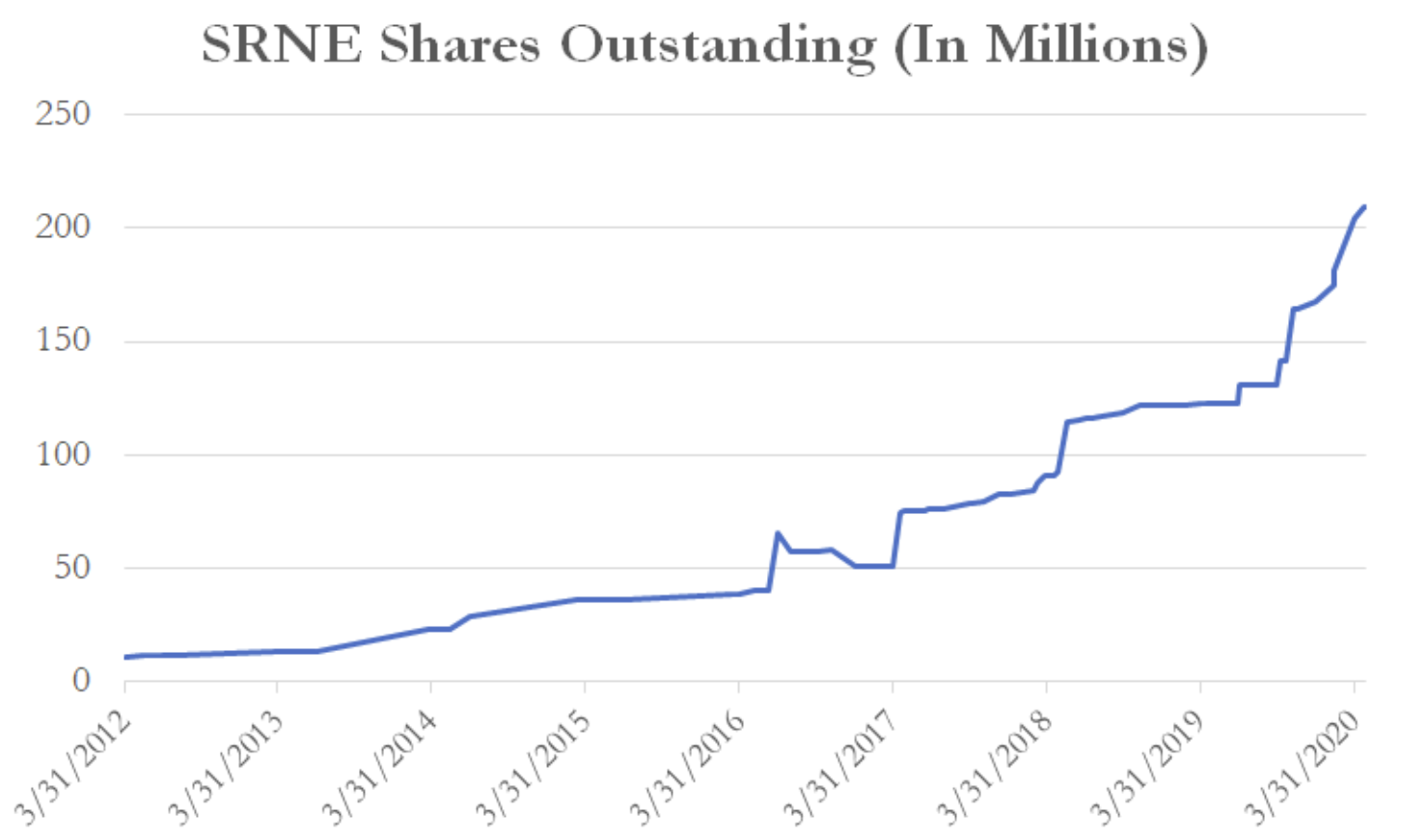
(Source: YCharts shares outstanding data)
By April 27th, the company had entered into 2 agreements to sell its stock on the open market, collectively calling for the company to sell up to $500 million in stock. It seemed like an absurdly optimistic number at the time, given that the entire market capitalization of the company was only $464 million.
One agreement was with A.G.P./Alliance Global Partners, a brokerage firm known for small cap PIPE equity financings.
One Financing Agreement Calls for Selling $250 Million in Stock Through a Mystery Financier in the British Virgin Islands. We Linked This Financier To DryShips, Which Suffered A Spectacular 99.9% Share Price Decline In The Year It Was Financed.
The other agreement was with a discrete entity called Arnaki Ltd., which BVI Corporate Records show is just over a year old. The agreement lists the name of a director of Arnaki, but we have learned from multiple industry sources that the key unnamed backer of Arnaki is actually an individual named Marc Bistricer.
Bistricer is a Canadian financier who runs a firm called Murchison Capital and has reputation for backing companies that have had sudden stock spikes followed by dramatic falls. One such company was DryShips.
DryShips fell 99.9% in 2017 [1,2] before later being acquired at $5.25 per share in 2019. The company’s split-adjusted all time high was over $70,000,000 per share, owing to eight reverse splits[1] in 2016 and 2017. The company subjected its shareholders to a seemingly constant state of equity dilution.
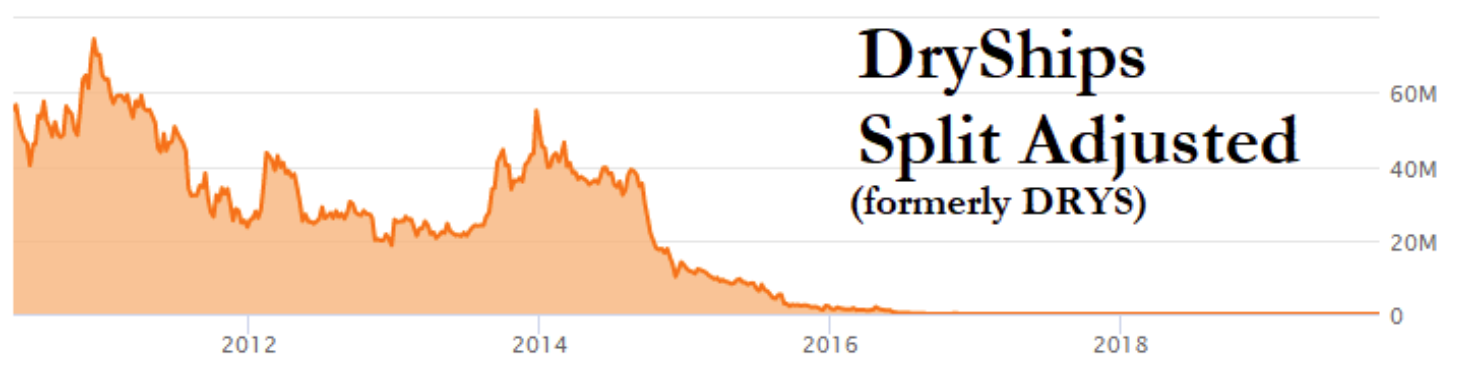
(Source: FactSet pricing data)
The financier behind DryShips was a mystery BVI entity called Kalani Investments, and its unknown backer drew widespread questions at the time.

Following the collapse of the equity, the entity was later revealed to be backed by Bistricer:

When most promising biotech companies need capital, they often use reputable investment banks to place the funding and generally seek institutional or strategic investment capital.
Instead of that typical route, Sorrento instead appears to be choosing to sell at-the-market (ATM) offerings directly to its largely retail investor base via an opaque BVI-based entity backed by Bistricer. We think this bodes poorly for investors.
We have reached out to Bistricer and Sorrento for comment and to confirm the relationship, and have yet to receive a reply as of this writing.
The Company Announced a Partnership With Mt. Sinai, Then “Blitzed” Journalists With A “Very Disingenuous” “Nonsense News” Story About A Cure Three Business Days Later
With its financing arrangements in place, the company moved at a rapid pace.
On Friday May 8th, the company announced a partnership with Mount Sinai, and that the two organizations “agreed to join forces in the investigation and development of an antibody cocktail”.
By Tuesday May 12th—a mere 3 business days later—Sorrento began pitching journalists an embargoed story with the email entitled “Sorrento discovers 100% COVID 19-Blocking Antibody” according to veteran biotech journalist Adam Feuerstein.

With apparently no other outlets taking the bait, Fox ran with the “exclusive” at the end of the week, on Friday May 15th.
Feuerstein, who is the Senior Writer for STAT News and is widely respected in the biotech space, said on Twitter that he didn’t take the “nonsense” story because he thought the pitch was “dumb”.
Adam Crisafulli of Vital Knowledge Media followed suit, calling Sorrento’s statements “very disingenuous”.
Sorrento Boldly Claims To Fox News: “There Is A Cure” and “If We Have The Neutralizing Antibody In Your Body, You Don’t Need The Social Distancing.”
On the morning of Friday, May 15, Sorrento probably garnered the most worldwide attention the company has ever received. Fox News published what looked like incredible news for the global fight against coronavirus.
Sorrento had reportedly made an “antibody breakthrough” in the fight against Covid-19, discovering an antibody that could “flush” Covid-19 out of a person’s system “within four days”, the article claimed.
The company said its antibody can provide “100% inhibition” of COVID-19, adding that a treatment could be available months before a vaccine hits the market.
The article was laden with quotes from management that made it seem like a Covid-19 treatment could be imminent. For example, a senior Vice President is quoted in the article as saying:
“As soon as it is infused, that patient is now immune to the disease,” Dr. Brunswick said to Fox News. “For the length of time, the antibody is in that system. So, if we were approved [by the FDA] today, everyone who gets that antibody can go back to work and have no fear of catching COVID-19.”
Sorrento CEO Dr. Henry H. Ji further claimed:
“When the antibody prevents a virus from entering a human cell, the virus cannot survive,” Dr. Ji said. “If they cannot get into the cell, they cannot replicate. So it means that if we prevent the virus from getting the cell, the virus eventually dies out. The body clears out that virus.”
“This puts its arms around the virus. It wraps around the virus and moves them out of the body.”
The CEO appealed to the senses of everyone struggling with life under quarantine, painting a picture of life going back to normal:
“If we have the neutralizing antibody in your body, you don’t need the social distancing. You can open up a society without fear.”
Reality Check: Former C-Suite Sorrento Executive: I Would “Never In My Life Put Out A Press Release Where I Say We Have A Cure”, There Are “Literally Hundreds” Of R&D Groups With Antibodies Similar To Sorrento
Given the optics of the article – namely that it appeared Sorrento was stating that they had a cure for the virus, we undertook some due diligence to make sure that Fox Viewers and investors in the company’s stock weren’t just getting one side of the coin.
We reached out to former employees at Sorrento and were able to speak with a former C-suite executive, who worked at the company for years and has followed its developments closely.
He immediately started by pointing out the company’s capital needs:
“I’m sure if you’re considering the company as an investment you have now looked at their balance sheet and you have looked at their … cash needs. Let’s say the Q1 2020 data, the numbers indicate, a somewhat precarious situation…”
When asked to comment on the antibody that Sorrento found, he explained to us that there are “literally hundreds” of R&D groups that have something similar:
“If you now identify an antibody that binds to an epitope of the viral spike protein, and you thereby inhibit that virus to bind to the ACE-2 entry receptor on the human cell and you thereby inhibit infection, my loose estimate would be that there are literally hundreds of R&D groups that have something like that.
There’s a European consortium that is comprised of over 130 academic groups funded by the European union, there’s a German university in there who have an activating antibody, there’s Regeneron who think that they can actually have a product available by Q3, there’s Amgen, there Lily and at this point in time the technologies to create a reasonably high affinity antibody against a viral epitope – they’re so mature that you simply have to assume there are literally hundreds of groups that have that..”
He was also the one of many experts we talked to who warned us about making such bold claims with in vitro results:
“…you do know that when somebody has data from an agent that shows efficacy in an in vitro assay – that if you then say I have now a cure, then this statement can only be understood narrowly…”
Finally, he made it extremely clear that he would not have put out the release in the way that Sorrento did:
“I’ve been [executive] of (multiple) public companies, I would have never in my life issued a press release where – based on a feasibility experiment I would at best say I have a lead molecule – issue a press release where I say we have a cure. To say that that’s outside of convention is probably the nicest way to put it.”
More Former Sorrento Employees: “I Suggest Extreme Skepticism Regarding Any Claims Made by Sorrento” And “The Announced Timeline Seems Very Aggressive”
We reached out to another former company VP, who worked at Sorrento recently for about a year, with our concerns about the company’s recent news.
He told us that he thought our “instincts were probably correct” and then simply said:
“I suggest extreme skepticism regarding any claims made by Sorrento.”
Yet another former Sorrento employee, who used to be a senior scientist at the company for more than six years, told us that while they thought the results looked promising, “they are only in-vitro data.” This former employee then warned:
“It is too early to really tell if it will translate in vivo. And we would have to see safety data. Also, their announced timeline seems very aggressive…”
Healthcare Experts & Mt. Sinai Department Of Medicine And Microbiology Workers: “This Looks Very Hyped” and “Nothing In Medicine Is 100%”
The Fox News article also noted that “Sorrento had partnered with New York-based health care system Mount Sinai to develop an antibody cocktail” for fighting coronavirus.
Knowing that Mount Sinai is a reputable name in the healthcare community, we wanted to reach out to them and see if it shared the same imminent optimism as it appeared Sorrento did.
When reached via e-mail about the Fox News article, one researcher at the Department of Medicine and Microbiology at Mt. Sinai told us:
“This looks very hyped. You need massive amounts of antibody to achieve this. This is the reason why this is not used for influenza. Too expensive, too much antibody needed. This cannot be a solution for everybody. There are no data yet in humans. For Ebola, there were several antibodies that worked like this one in vitro, but only a few are protective in vivo. Bottom line, very early in development to know feasibility.”
A second Mt. Sinai research worker that we reached via e-mail simply told us that:
“In general terms…nothing in medicine is 100%. Nothing.”
We also communicated with Dr. Charles Rice, the Chair in Virology & Head of the Laboratory of Virology and Infectious Disease at Rockefeller University, who told us:
“I don’t know the details of the Sorrento MAb but their claims at this apparent stage of development, without clinical data, seem overstated. There are dozens of groups developing these antibodies and time (and appropriate tests) will tell which are most effective. As a general solution or “cure” it is unlikely that an infused product, even if long lasting, will cover all of the bases needed to control this infection.”
Finally, a PhD at the National Institute of health warned us:
“…be cognizant of the stage of the research (ex. if there is only data in vitro, which means in a petri dish).”
Regeneron Co-Founder and Chief Scientific Officer: “They Seemingly Are Substantially Behind the Leaders At This Time…It is Very Hard to Seriously Evaluate the Sorrento Effort”
We also communicated with George Yancopoulos, Co-Founder, President & Chief Scientific Officer for Regeneron, which itself aims to advance an antibody treatment for COVID-19. His statements were candid in describing the competitive field that Sorrento and his company faced, and the challenges in bringing an antibody treatment to market:
“Several companies have previously announced that they have already generated very potent anti-viral neutralizing antibodies – including our company as we announced a while back. That is only the first step, and there are many more steps to manufacture and progress such antibodies into clinical trials – I believe most accounts suggest that our company and probably one other company (Lilly) are the leaders into progressing these antibodies into clinical trials, and both of us are planning on doing this in the next few weeks. And then there are the challenges of successfully carrying out these clinical trials, which is also not guaranteed.
I am not aware of any data suggesting Sorrento’s single Ab is as good as the many others that had already been previously generated (such as ours or Lilly’s), nor that they have any of the required downstream capabilities, nor that they have demonstrated any capabilities or success previously in other programs (such as the success we demonstrated with our related efforts against Ebola), and they seemingly are substantially behind the leaders at this time.
So it is very hard to seriously evaluate the Sorrento effort.”
This Doesn’t Appear to Be Sorrento’s First Questionable Stock Market Surprise—A Series of Suspicious Anonymous Buyout Offers
Sorrento has a history of eyebrow raising announcements that happen to positively influence its stock price.
On November 22nd 2019, the company filed a form S-3 seeking to register over 17 million shares of stock that had been newly converted from holders of promissory notes earlier in the month. [Pg. 2]
Three days later, on November 25th, the company announced that it had received and summarily rejected a “non-binding” offer by two anonymous biopharmaceutical companies. The companies proposed acquiring the stock for between $3.00 and $5.00 per share.
The stock jumped 94% on the news, to $3.11, from a prior-day closing price of $1.60, on almost 72x normal volume.
The registration statement for the 17 million shares had not yet become effective however, meaning the shareholders were unable to sell.
Thankfully for them, on January 10th 2020, the company announced that it had received yet another non-binding acquisition proposal from an unnamed private equity fund, offering up to $7 per share. The news sent the stock to $4.76, up ~40% from its previous day’s close, on ~7x its average volume.
The registration statement for the 17 million shares became effective on January 23rd, granting the financiers the ability to unload their stock. It is unclear from filings whether they chose to do so. On January 27th, the company announced that it had rejected the latest anonymous offer. From there, the stock faded into the low $2 range in the lead-up to the COVID-19 ‘cure’ series of events.
We asked the company who the anonymous private equity fund was, and have not yet received a reply.
We have decades of combined public market investing experience and have never seen an anonymous bidding war for a company. Given the proximity to the company’s equity issuances we find the series of events to yet another troubling red flag.
Conclusion: When It Seems Too Good To Be True…
As we have urged with other names participating in the Covid-19 news cycle, we believe it’s worth remaining cautious on Sorrento and its suggested coronavirus cure.
We also believe that given the egregious nature of how the company sought media attention and the way it framed the story – combined with the fact that the company was already prepared to raise hundreds of millions of dollars in advance of finding its supposed cure – that regulators should carefully scrutinize the company’s actions.
Disclosure: We are short shares of Sorrento (NASDAQ:SRNE)
Legal Disclaimer
Use of Hindenburg Research’s research is at your own risk. In no event should Hindenburg Research or any affiliated party be liable for any direct or indirect trading losses caused by any information in this report. You further agree to do your own research and due diligence, consult your own financial, legal, and tax advisors before making any investment decision with respect to transacting in any securities covered herein. You should assume that as of the publication date of any short-biased report or letter, Hindenburg Research (possibly along with or through our members, partners, affiliates, employees, and/or consultants) along with our clients and/or investors has a short position in all stocks (and/or options of the stock) covered herein, and therefore stands to realize significant gains in the event that the price of any stock covered herein declines. Following publication of any report or letter, we intend to continue transacting in the securities covered herein, and we may be long, short, or neutral at any time hereafter regardless of our initial recommendation, conclusions, or opinions. This is not an offer to sell or a solicitation of an offer to buy any security, nor shall any security be offered or sold to any person, in any jurisdiction in which such offer would be unlawful under the securities laws of such jurisdiction. Hindenburg Research is not registered as an investment advisor in the United States or have similar registration in any other jurisdiction. To the best of our ability and belief, all information contained herein is accurate and reliable, and has been obtained from public sources we believe to be accurate and reliable, and who are not insiders or connected persons of the stock covered herein or who may otherwise owe any fiduciary duty or duty of confidentiality to the issuer. However, such information is presented “as is,” without warranty of any kind – whether express or implied. Hindenburg Research makes no representation, express or implied, as to the accuracy, timeliness, or completeness of any such information or with regard to the results to be obtained from its use. All expressions of opinion are subject to change without notice, and Hindenburg Research does not undertake to update or supplement this report or any of the information contained herein.
[1] 03/11/2016 1 for 25, 08/15/2016 1 for 4, 11/01/2016 1 for 15, 01/23/2017 1 for 8, 04/11/2017 1 for 4, 05/11/2017 1 for 7, 06/22/2017 1 for 5 and 07/21/2017 1 for 7
33 thoughts on “Sorrento’s Pandemic Profiteering: Experts and Former Employees Speak Out on Sensational Claims of Covid-19 Cure”
Comments are closed.
Nice article.
Excellent research from you and Viceroy! Just wish I’d been able to short more.
Thanks for your superb reporting!
thank you very much for your efforts a magnomity. you provide a most valuable comodity its called insite. if some amiricans had insite we wouldnt be where we are now.
cheers Tom
Once again, another pathetic and not surprising effort to drag down a good company, doing good work for real people. I cannot believe people actually trade on your BS. You write like a high school sophomore and rely solely on misrepresentation and half-truths. God forbid you’d actually do some analysis! It’s like your the Donald Trump of stock analysis. Good luck short pants.
Agreed
agreed ! what come around go around KARMA will get to them
Hindenburg = Nathan Andrews right? Anyhow, quoting unnamed scorned ex-employees is hardly groundbreaking news. You are well known to write hit pieces full of inaccuracies and omissions to better your short position and make money. Nothing you’ve claimed can be legitamtely confirmed or corroborated and I can’t wait for a jury to hear all of this and render a decision.
It is time you are held accountable because this is not an isolated incident.
Let’s see your advanced analysis, you pretentious twat. You have not addressed a single observation or argument from the author, so your insults carry zero weight.
We will see how the short sellers fair when the news about the antibody is legit and we have some 300 to 400 million share days as I will be one buying as many shares as I can get my hands on to a minimum of 10 dollars. If I was Sorrento, I would sue!
Good luck shorties! Bulls against bears with the market moving back up to pre-covid levels!
Is there a possibility of you being sued for false reporting?
Iguess you could also-be sued for slander and false allegations. Why would former employees speak out, I would venture to guess they have a non disclosure contract that is still in affect
Hindenburg research is just trying to ensure they cover their short position. Looks desperate to me, the floor is at 5 dollars anyway. Here’s the thing, SRNE is the industry leader in antibody research and developement. LEADER. That means they have the competitive edge to produce results. Put that in your pipe and smoke it
I find the report very informative, and indeed, it is consistent with many red flags that can be found on Sorrento’s financials. From their annual report:
https://investors.sorrentotherapeutics.com/static-files/c82f1fb8-3ab2-4249-a733-acaf6591532a
Check out page 108 of their financials:
2018 Debt to Equity Ratio: 2.01
2019 Debt to Equity Ratio: 16.02 (!!!)
They have since cut the debt down according to recent announcements, but where did the money come from to do this?
Now scroll down to near the bottom of the document, Exhibit 21.1. Notice that Sorrento has *12* subsidiaries, with 3 located in China. Most large corporations don’t even have 12 subs, much less a small cap. biotech firm. Why does it have so many, and 3 in China? This is a complex rabbit hole, which seems designed to obfuscate money flow. We are in the middle of a trade war with China, and China is considered a serious economic adversary of the US. This company is in bed with China. Does the Chinese government have its hand in it? This is quite possible, even probable.
According to the 1-K the the business in China is manufacturing. They also manufacture CBD oil internationally as there are issues with manufacturing it here in the USA. I suggest doing a bit more research before spreading out erroneous speculation and irresponsible disinformation.
You have ZERO idea of what you are talking about! Don’t post conjecture!
Hope this performs as well or better than GSX did after your short call. That stock has run from $30 to $63! You talk about the company citing anonymous sources but you yourself rely on anonymous tips from “former employees” and medical professionals.
STRONG BUY! This is not about the therapy at this time anyway, FDA EUA for 8 minute COVID testing kits runs this to teens.
Happy 4th!
Not really. none of the sources can be confirmed this could just all be BS. likely is
why is this guy is not in jail yet, that is what im wondering
Interesting to see what happens with this one… maybe just another company taking advantage or maybe they really have it.